Introduction
Progressive weakness occurs infrequently in patients with diabetic polyneuropathy (DPN), and its cause needs to be carefully differentiated. However, the differential diagnosis is often complicated by preexisting abnormalities that affect physical examination and nerve conduction studies (NCS).
Amyotrophic lateral sclerosis (ALS) is a degenerative disease of motor neurons characterized by slowly progressive asymmetric weakness that does not respond to treatment. ALS can be diagnosed when upper motor neuron (UMN) and lower motor neuron abnormalities are observed, sensory symptoms are absent, and other possible diseases have been ruled out [
1]. However, patients with ALS and DPN may not show UMN abnormalities and may have abnormalities on sensory NCS.
In chronic inflammatory demyelinating polyradiculoneuropathy (CIDP), paresthesia and muscle weakness gradually progress or relapse and remit repeatedly. Unlike ALS, 60% to 90% of patients with CIDP show improvement with immunomodulatory therapy [
2]. However, when CIDP develops in patients with DPN, the clinical features and abnormalities of the NCS may be obscured, making it difficult to differentiate between the two diseases. CIDP should be considered if demyelinating features, such as decreased nerve conduction velocity, conduction block, and temporal dispersion, are present on NCS [
3].
The authors report a patient with CIDP who was initially misdiagnosed with ALS due to progressive weakness while being treated for diabetes, but showed demyelinating features on NCS, and improved after continuous high-intensity immunomodulatory therapy.
Case Report
A 64-year-old woman presented with progressive limb weakness. In October 2018, the patient began experiencing hypesthesia in both feet and decreased muscle strength in both lower extremities. In July 2019, she experienced difficulty performing fine motor movements such as fastening buttons or closing bottle caps owing to weakness in both hands. She had been diagnosed with hypertension and diabetes mellitus 7 years prior and was taking medication. NCS showed polyneuropathy (
Tables 1,
2) and electromyography revealed a wide range of denervation potentials (
Table 3) at that time. She was initially diagnosed with ALS and DPN. However, another doctor diagnosed her with CIDP based on the results of NCS—namely, conduction block, temporal dispersion, and a sural-sparing pattern—and treated her with oral prednisolone (10 mg/day) and azathioprine (100 mg/day). Unfortunately, even after medical treatment, muscle weakness progressed to the proximal arms and legs, and neuropathic pain in the extremities worsened. As an additional treatment, intravenous immunoglobulin (IVIg) was injected at a dose of 0.4 g/kg once every 2 weeks. After IVIg treatment, neuropathic pain subsided slightly, but muscle weakness progressed. She visited Chungnam National University Hospital in March 2020.
On a neurological examination, she could not sense vibrations in both feet. A muscle examination using the Medical Research Council (MRC) score showed bilateral shoulder abduction 4/4, bilateral elbow flexion and bilateral wrist extension 4/4, bilateral finger flexion 3/3, bilateral finger extension 1/1, bilateral hip flexion 3/3, bilateral knee extension 4/4, and bilateral ankle flexion 0/0. The MRC sum score (MRCSS) was 38, indicating severe distal muscle weakness (
Fig. 1) [
4]. The deep tendon reflexes were decreased, and there were no UMN abnormalities. In the cerebrospinal fluid (CSF) test, the total protein level was elevated to 118 mg/dL without an increase in leukocytes.
In the NCS, the amplitude of the compound muscle action potential and nerve conduction velocity were further reduced compared to the previous test, and conduction block and temporal dispersion were more definite (
Fig. 2). This was compatible with CIDP according to the electrical diagnostic criteria of the European Federation of Neurological Societies/Peripheral Nerve Society (EFNS/PNS) presented in 2021 (
Tables 1,
2) [
2]. Normal findings were observed for anti-ganglioside antibodies, anti-myelin-associated glycoprotein (anti-MAG) antibody, and immunofixation testing of serum and urine samples. These findings led us to rule out multifocal motor neuropathy in diabetes and anti-MAG neuropathy. Although the patient’s NCS findings met the electrical diagnostic criteria for CIDP, the differential diagnosis needed to consider autoimmune nodopathy and late-onset hereditary neuropathy because the symptoms progressed very slowly and did not respond to standard immunomodulatory therapy. Autoimmune nodopathy testing using paranodal antibodies such as neurofascin155 (NF155) and contactin-associated protein-1 (Caspr-1) was not available when the patient initially presented, and nodopathy testing began for research purposes in the Korean context after the patient had received immunomodulatory therapy. The patient’s symptoms improved after therapy, so we were unable to perform the test subsequently. Furthermore, late-onset hereditary polyneuropathy could be ruled out because it does not improve with IVIg treatment and does not worsen after IVIg discontinuation.
A biopsy of the left superficial peroneal nerve revealed no specific findings except the infiltration of a few inflammatory cells (
Fig. 3). Based on the test results, the patient was diagnosed with CIDP that was not sufficiently treated; a high-dose steroid (1,000 mg/day) was injected intravenously for 3 days starting on April 2, 2020, and prednisolone (10 mg/day) and azathioprine (100 mg/day) were maintained. However, muscle strength deteriorated even after high-dose steroid injection treatment; thus, IVIg (0.4 g/kg/day) was administered for 5 days starting on April 18, 2020, and then maintained at a dose of 1 g/kg/day at 2-week intervals. With continuous treatment, the symptoms began to improve gradually. In January 2022, a muscle examination showed bilateral finger extension 4/4 and bilateral ankle flexion 3/3, and the MRCSS score was 50 (
Fig. 1). The NCS also improved compared to March 2020 (
Tables 1,
2). Thus, the IVIg treatment was adjusted to 3-week intervals from January 2022 onwards; thereafter, muscle strength gradually deteriorated, and she could not walk independently. In June 2022, IVIg was re-adjusted to 2-week intervals. Nevertheless, the symptoms continued without worsening or improvement, and the results of NCS were aggravated in September 2022 (
Tables 1,
2). In March 2023, IVIg was discontinued due to an insufficient effect, and rituximab (1,000 mg/day) started to be administered every 2 weeks. After the discontinuation of IVIg, muscle weakness suddenly worsened, and the MRCSS decreased to 18. Upon restarting IVIg every 2 weeks, her muscle weakness has been improving rapidly (
Fig. 1).
The need for informed consent was waived due to retrospective nature of the case.
Discussion
ALS involves progressive muscle weakness, with a wide range of denervation potentials observed on electromyography. However, the patient was diagnosed with CIDP based on nerve conduction tests revealing demyelination and a fluctuating clinical course according to immunomodulatory treatment. Furthermore, in demyelinating polyneuropathy, abundant spontaneous activity can be observed due to secondary axonal changes. Additionally, a sural-sparing pattern shown on initial sensory NCS is suggestive of CIDP rather than DPN.
Some cases have been reported of patients with progressive muscle weakness as the main symptom, who were initially diagnosed with CIDP and treated accordingly, but were eventually confirmed as having ALS [
5,
6]. These cases showed demyelinating abnormalities in motor and sensory NCS, and the symptoms gradually progressed from 1 to 2 years. None of these patients responded to IVIg or steroid treatment. In some cases, a peripheral nerve biopsy showed demyelinating neuropathy, an autopsy showed corticospinal tract degeneration, and a pathological examination showed both peripheral neuropathy and abnormal findings, suggestive of ALS. However, whether peripheral neuropathy was caused by ALS or was coincidental in these cases remained unconfirmed. Unlike previous cases, our patient showed muscle strength improvement with continuous IVIg and steroid treatment, and her muscle strength worsened after stopping IVIg treatment and improved again with rituximab and immunoglobulin treatment. Therefore, this patient clearly responded to immunomodulatory therapy, and it was reasonable to diagnose her with CIDP because the EFNS/PNS diagnostic criteria had been satisfied [
2].
The diagnostic criteria presented in 2010 by the EFNS/PNS are the most widely used for diagnosing CIDP [
7]. The EFNS/PNS recently announced revised CIDP criteria based on a combination of expert opinions [
2]. If CIDP is suspected, the diagnosis is based on clinical features, NCS results, CSF test results, nerve biopsy results, and treatment response. In the present case, the initial NCS did not meet the electrical diagnostic criteria for CIDP. Upon visiting our hospital, both the clinical features and the NCS met the EFNS/PNS diagnostic criteria, making the diagnosis of CIDP possible.
CIDP often occurs in conjunction with diabetes or monoclonal gammopathy; however, the relationship between these diseases has not yet been clearly identified. The risk of CIDP is reportedly higher in individuals with diabetes [
8,
9]. As paresthesia is the main symptom of DPN, the possibility of other diseases must be considered if muscle weakness progresses rapidly or occurs asymmetrically. CIDP is likely if decreased nerve conduction velocity or demyelinating abnormalities, such as conduction block and temporal dispersion, are observed on NCS. DPN may be difficult to differentiate from CIDP because of similar patterns [
8,
9]. If DPN progresses, muscle weakness can occur, and a decrease in nerve conduction velocity can be observed. However, in DPN, the total CSF protein level does not rise above 100 mg/dL, and demyelinating abnormalities that meet the electrodiagnostic criteria for EFNS/PNS are rarely observed [
3].
In this case, CIDP was diagnosed and immunomodulatory treatment was administered at the first hospital. However, improvement was not observed owing to insufficient treatment. Currently, CIDP treatment includes immunomodulatory therapy, with corticosteroids, IVIg, and plasmapheresis being the main treatments. The EFNS/PNS diagnostic criteria include response to treatment as a supportive diagnostic criterion. If immunomodulatory therapy is effective, there are currently no guidelines for optimal maintenance therapy, and treatment should be maintained for an adequate period at a sufficient dose for each patient, depending on the patient’s condition [
10].
In patients with DPN and progressive muscle weakness, caution is required in the differential diagnosis. CIDP is suspected when demyelinating abnormalities and a sural-sparing pattern are observed on NCS, as in this case, and immunomodulatory treatment at a sufficient dose for a sufficient period is required.
Conflict of Interest
Eunhee Sohn is an editorial board member of the journal, but she was not involved in the peer reviewer selection, evaluation, or decision process of this article. No other potential conflicts of interest relevant to this article were reported.
Fig. 1.
Diagram of the patient’s clinical course and medical management. The Medical Research Council (MRC) sum score is the sum of the MRC scores of bilateral shoulder abduction, elbow flexion, wrist extension, hip flexion, knee extension, and ankle dorsiflexion, ranging from 0 to 60. The distal sum score is the sum of the MRC scores of bilateral ankle dorsiflexion, finger abduction, finger flexion, and finger extension, ranging from 0 to 40. IVIg, intravenous immunoglobulin.

Fig. 2.
Waveforms of nerve conduction studies. Initial nerve conduction studies of the right median nerve (A), right ulnar nerve (B), right tibial nerve (C), and left tibial nerve (D). (E, F, G, H) Second nerve conduction studies. Conduction block and temporal dispersion were more definite.
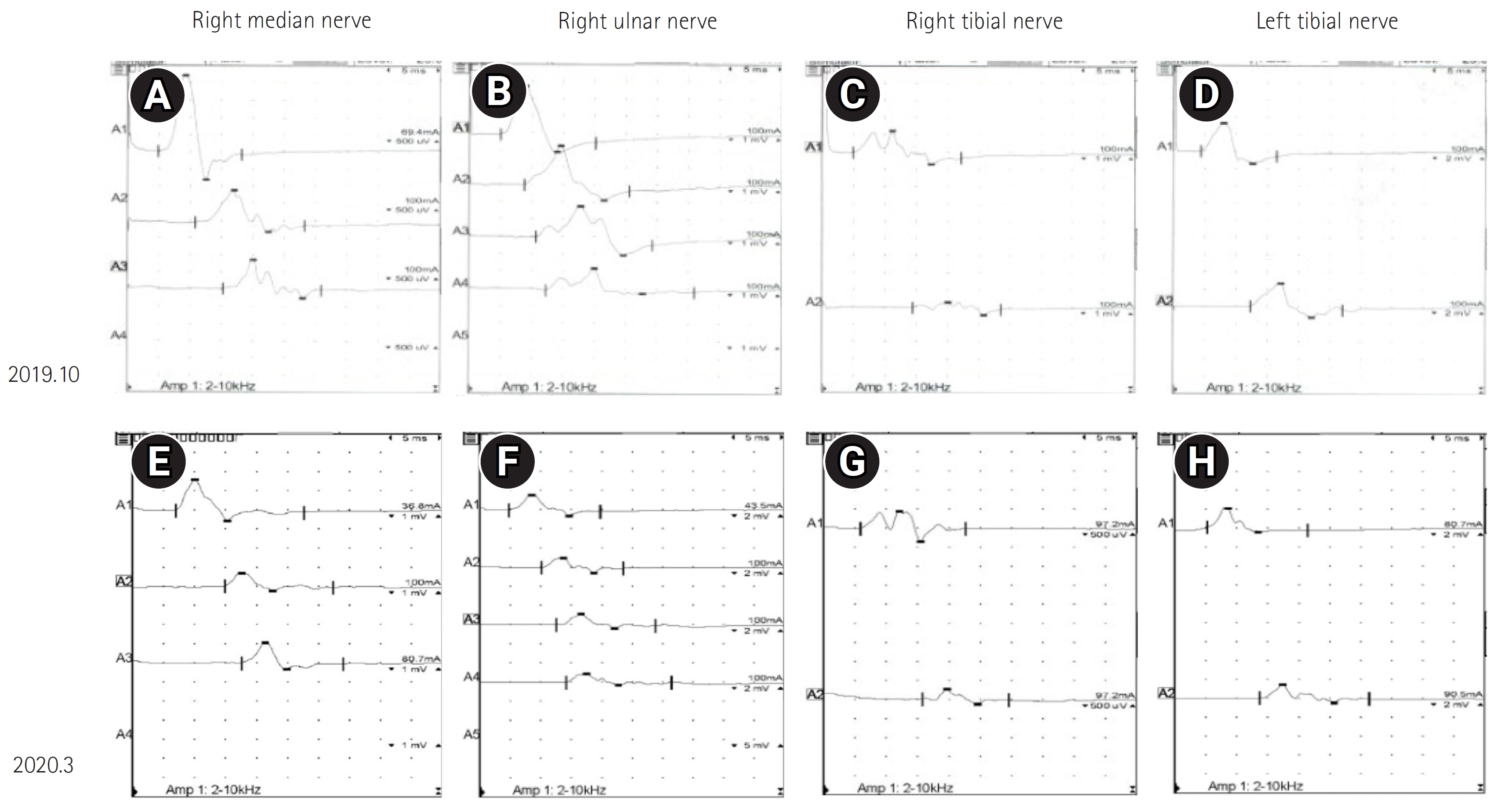
Fig. 3.
Histopathological and immunohistochemical examination of superficial peroneal nerve biopsy. (A) A few T cells positive for cluster of differentiation 3 (CD3) protein (×200 magnification), (B) a few mononuclear cells on H&E stain (×200 magnification), (C) myelin fragmentation on H&E stain (×200 magnification). The pathological findings show a few cells with positivity for CD3 and B-cell lymphoma 2 (Bcl-2). (D, E) Yellow arrows show nonspecific myelin collapse on electron microscopic examination.

Table 1.
Motor Nerve Conduction Studies
|
Variable |
Oct 2019 |
Mar 2020 |
Sep 2021 |
Sep 2022 |
|
Latency (ms) |
Amp (mV) |
NCV (m/sec) |
Latency (ms) |
Amp (mV) |
NCV (m/sec) |
Latency (ms) |
Amp (mV) |
NCV (m/sec) |
Latency (ms) |
Amp (mV) |
NCV (m/sec) |
|
Median (R/L) |
|
|
|
|
|
|
|
|
|
|
|
|
|
APB-W |
4.8*/7.4*
|
3.1*/1.8*
|
|
6.9*/8.0*
|
2.2*/1.2*
|
|
6.1*/6.7*
|
1.2*/1.9*
|
|
7.0*/7.5*
|
1.1/3.6*
|
|
|
W-E |
|
1.2*/0.7*
|
33*/38*
|
|
0.9*/0.2*
|
23*/31*
|
|
0.4*/1.3*
|
32*/32*
|
|
0.3*/2.0*
|
19*/29*
|
|
E-A |
|
1.1*/0.7*
|
28*/48*
|
|
|
|
|
|
|
|
0.2*/1.7*
|
61/58 |
|
Ulnar (R/L) |
|
|
|
|
|
|
|
|
|
|
|
|
|
ADM-W |
4.9*/4.1*
|
3.9*/3.3*
|
|
4.7*/6.4*
|
2.3*/1.8*
|
|
4.8*/4.8*
|
2.6*/2.3*
|
|
5.2*/5.5*
|
3.7*/2.8*
|
|
|
W-E |
|
3.2*/2.6*
|
51/50*
|
|
1.6*/1.3*
|
37*/34*
|
|
1.8*/2.0*
|
40*/31*
|
|
2.3*/1.1*
|
37*/21*
|
|
E |
|
2.9*/2.5*
|
50*/50*
|
|
1.5*/0.4*
|
42*/53 |
|
1.8*/2.1*
|
67/33*
|
|
2.1*/1.1*
|
40*/28*
|
|
A |
|
1.5*/2.5*
|
62/57 |
|
1.3*0.3*
|
63/67 |
|
1.9*/2.1*
|
73/57 |
|
2.1*/1.1*
|
57/53 |
|
Tibial (R/L) |
|
|
|
|
|
|
|
|
|
|
|
|
|
AH-Ak |
4.9/4.3 |
1.9*/4.8*
|
|
5.8*/5.2*
|
0.8*/2.6*
|
|
4.3/4.0 |
2.7*/3.7*
|
|
4.8/4.7 |
0.7*/3.2*
|
|
|
Ak-P |
|
0.8*/4.0*
|
32*/40*
|
|
0.4*/1.9*
|
33*/39*
|
|
1.5*/2.4*
|
41/46 |
|
0.7*/2.4*
|
41/42 |
|
Peroneal (R/L) |
|
|
|
|
|
|
|
|
|
|
|
|
|
EDB-Ak |
5.2*/5.1*
|
1.1*/1.4*
|
|
5.1*/5.7*
|
2.9*/2.0*
|
|
4.8*/4.8*
|
2.5*/2.9*
|
|
5.3*/5.6*
|
2.8*/1.5*
|
|
|
Ak-BF |
|
0.8*/1.5*
|
44/42 |
|
2.3*/1.5*
|
36*/35*
|
|
2.0*/2.1*
|
39*/34*
|
|
1.7*/0.7*
|
36*/33*
|
|
BF-AF |
|
NR*/NR*
|
NR*/NR*
|
|
2.4*/1.5*
|
64/69 |
|
2.1*/2.1*
|
69/56 |
|
1.7*/0.7*
|
41/41 |
Table 2.
Sensory Nerve Conduction Studies
|
Variable |
Oct 2019 |
Mar 2020 |
Sep 2021 |
Sep 2022 |
|
Amp (μV) |
NCV (m/sec) |
Amp (μV) |
NCV (m/sec) |
Amp (μV) |
NCV (m/sec) |
Amp (μV) |
NCV (m/sec) |
|
Median (R/L) |
|
|
|
|
|
|
|
|
|
F-W |
10.6/6.8*
|
44/39*
|
17/7*
|
38*/37*
|
12/10 |
39*/34*
|
18/10 |
37*/36*
|
|
W-E |
16.0/10.7 |
55/48*
|
15/12 |
48*/47*
|
13/14 |
47*/47*
|
14/13 |
53/54 |
|
E-A |
11.6/25.9 |
61/59 |
31/17 |
63/58 |
30/27 |
58/55 |
26/23 |
69/63 |
|
Ulnar (R/L) |
|
|
|
|
|
|
|
|
|
F-W |
17.8/9.5 |
50/45*
|
11/8 |
38*/46 |
11/10 |
46/41 |
11/8*
|
43/41 |
|
W-E |
17.4/7.4*
|
58/55 |
17/15 |
46*/53 |
19/16 |
54/54 |
14/13 |
48/48 |
|
E-A |
16.9/7.1*
|
56/57 |
23/14 |
63/65 |
19/24 |
72/76 |
12/10 |
59/62 |
|
Superficial peroneal (R/L) |
13.0/12.7 |
50/50 |
10/9 |
56/57 |
9/7 |
48/45 |
6/4 |
47/46 |
|
Sural (R/L) |
11.4/12.7 |
36/41 |
13/10 |
39/40 |
11/5*
|
40/45 |
7*/5*
|
42/42 |
Table 3.
|
Variable |
Insertional activity |
Spontaneous activity |
Volitional MUAPs |
Recruitment |
Interference |
|
Fib |
PSW |
Dur |
Amp |
Poly |
|
Oct 2019 |
|
|
|
|
|
|
|
|
|
PT |
Increased |
1+ |
1+ |
Normal |
Normal |
None |
Normal |
Normal |
|
EIP |
Increased |
1+ |
1+ |
Increased |
Large |
Poly |
Reduced |
Reduced |
|
FDI |
Increased |
1+ |
1+ |
Normal |
Normal |
None |
Normal |
Normal |
|
TA |
Increased |
3+ |
3+ |
Increased |
Large |
Poly |
Reduced |
Reduced |
|
GN med |
Increased |
3+ |
3+ |
Increased |
Large |
Poly |
Reduced |
Reduced |
|
C paraspinal |
Increased |
3+ |
3+ |
|
|
|
|
|
|
T paraspinal |
Increased |
1+ |
3+ |
|
|
|
|
|
|
LS paraspinal |
Increased |
2+ |
2+ |
|
|
|
|
|
|
Sep 2021 |
|
|
|
|
|
|
|
|
|
Biceps brachii |
Normal |
None |
None |
Increased |
Large |
Poly |
Reduced |
Reduced |
|
FDI |
Increased |
1+ |
1+ |
Increased |
Large |
Poly |
Reduced |
Reduced |
|
TA |
Increased |
1+ |
1+ |
Increased |
Large |
Poly |
Reduced |
Reduced |
|
GN med |
Normal |
None |
None |
Increased |
Large |
Poly |
Reduced |
Reduced |
|
Rectus abdominis |
Normal |
None |
None |
Normal |
Normal |
None |
Normal |
Normal |
|
C paraspinal |
Normal |
None |
None |
|
|
|
|
|
|
T paraspinal |
Normal |
None |
None |
|
|
|
|
|
|
LS paraspinal |
Increased |
1+ |
1+ |
|
|
|
|
|
References
1. Brooks BR, Miller RG, Swash M, Munsat TL; World Federation of Neurology Research Group on Motor Neuron Diseases: El Escorial revisited: revised criteria for the diagnosis of amyotrophic lateral sclerosis. Amyotroph Lateral Scler Other Motor Neuron Disord 2000;1:293-299.


4. Kleyweg RP, van der Meché FG, Schmitz PI: Interobserver agreement in the assessment of muscle strength and functional abilities in Guillain-Barré syndrome. Muscle Nerve 1991;14:1103-1109.


5. Rajabally YA, Jacob S: Chronic inflammatory demyelinating polyneuropathy-like disorder associated with amyotrophic lateral sclerosis. Muscle Nerve 2008;38:855-860.


6. Echaniz-Laguna A, Degos B, Mohr M, Kessler R, Urban-Kraemer E, Tranchant C: A study of three patients with amyotrophic lateral sclerosis and a polyneuropathy resembling CIDP. Muscle Nerve 2006;33:356-362.


7. Van den Bergh PY, Hadden RD, Bouche P, Cornblath DR, Hahn A, Illa I, et al: European Federation of Neurological Societies/Peripheral Nerve Society guideline on management of chronic inflammatory demyelinating polyradiculoneuropathy: report of a joint task force of the European Federation of Neurological Societies and the Peripheral Nerve Society-first revision. Eur J Neurol 2010;17:356-363.


8. Rajabally YA, Peric S, Cobeljic M, Afzal S, Bozovic I, Palibrk A, et al: Chronic inflammatory demyelinating polyneuropathy associated with diabetes: a European multicentre comparative reappraisal. J Neurol Neurosurg Psychiatry 2020;91:1100-1104.


9. Doneddu PE, Cocito D, Manganelli F, Fazio R, Briani C, Filosto M, et al: Frequency of diabetes and other comorbidities in chronic inflammatory demyelinating polyradiculoneuropathy and their impact on clinical presentation and response to therapy. J Neurol Neurosurg Psychiatry 2020;91:1092-1099.


10. Bunschoten C, Jacobs BC, Van den Bergh PY, Cornblath DR, van Doorn PA: Progress in diagnosis and treatment of chronic inflammatory demyelinating polyradiculoneuropathy. Lancet Neurol 2019;18:784-794.














